Biotech

Aclarion Reports 196% Surge in Nociscan Scans, Advances Reimbursement and Clinical Trials
Aclarion's Q1 2026 results show a 196% year-over-year increase in Nociscan scan volumes, driven by clinical adoption and payer coverage, with a strong cash position and upcoming CLARITY trial data expected in late 2026.
TransCode Therapeutics Appoints Scientific Advisory Board Chair, Partners with Michigan State University to Advance RNA-Based Cancer Therapies
TransCode Therapeutics announced the appointment of Dr. Anna Moore as Chair of its Scientific Advisory Board and a sponsored research agreement with Michigan State University to advance its RNA-based therapeutic pipeline, including lead candidate TTX-MC138.

Oncotelic Therapeutics Featured in BioMedWire Editorial on Biotech Valuation Shift
Oncotelic Therapeutics' inclusion in a BioMedWire editorial highlights how advancing drug pipelines are reshaping biotech valuations, with its AI-driven oncology pipeline and 45% stake in GMP Bio serving as examples of innovation translating into tangible enterprise value.

Biotech Valuation Shift: Drug Pipeline Progress Now Measured as Financial Assets Under GAAP
The biotech sector is experiencing a transformation where advancing drug pipelines are increasingly valued as financial assets under fair-value accounting, with companies like Oncotelic Therapeutics exemplifying how clinical progress directly impacts balance sheet valuation.
Soligenix CEO Advocates for Patient-Centric Reformulation Strategy in Rare Disease Drug Development
Soligenix CEO Christopher Schaber argues that reformulating therapies to better fit patients' daily lives can improve access, adherence, and long-term viability, highlighting a strategic shift in biopharmaceutical development.

Oncotelic Therapeutics Featured in Editorial on Biotech Valuation Trends
Oncotelic Therapeutics' diversified pipeline and strategic holdings, including a 45% stake in GMP Bio valued at over $1 billion, exemplify how scientific progress and commercialization proximity are increasingly recognized as measurable financial assets under U.S. GAAP, reflecting a broader shift in biotech valuation.

Soligenix CEO Emphasizes Capital Discipline as Key to Biotech Success in Constrained Funding Environment
Soligenix CEO Dr. Christopher J. Schaber highlights capital discipline as essential for biotech companies to navigate today's challenging funding landscape, where strategic resource allocation and milestone-focused development are critical for attracting investment and advancing programs toward commercialization.

March Biosciences Receives FDA RMAT Designation for CAR-T Therapy Targeting Aggressive T-Cell Lymphoma
The FDA granted RMAT designation to March Biosciences' MB-105 CAR-T therapy for relapsed T-cell lymphoma, potentially accelerating treatment development for patients with limited survival options.

Soligenix Reports Q3 2025 Results with Key Clinical Milestones for Rare Disease Treatments
Soligenix Inc. announced positive clinical progress for its rare disease pipeline including upcoming Phase 2a psoriasis results and maintained safety profile for HyBryte™ in CTCL treatment, supported by sufficient funding through 2026.

Datavault AI Grants Scilex Exclusive License for Biotech Exchange Platform in $2.55 Billion Deal
Datavault AI has licensed its AI-driven technology to Scilex for a biotech exchange platform that could address a $2 trillion pharmaceutical market, representing a major advancement in tokenizing and monetizing healthcare assets.

Cybin Inc. Announces Q2 2025 Financial Results and Business Update Conference Call
Cybin Inc. will host a conference call on November 13, 2025, to discuss its second-quarter financial results and provide updates on its late-stage neuropsychiatry pipeline, including breakthrough-designated psychedelic treatments for major depressive disorder and generalized anxiety disorder.

GeoVax to Report Q3 2025 Financial Results and Provide Business Update
GeoVax Labs will report its third quarter 2025 financial results and provide updates on its clinical-stage vaccine and immunotherapy programs, including COVID-19 vaccines for immunocompromised patients and oncology treatments.

Soligenix Strengthens CTCL Advisory Board to Advance HyBryte Therapy Development
Soligenix has updated its U.S. Medical Advisory Board for cutaneous T-cell lymphoma to support the clinical advancement of HyBryte and related therapies, reflecting the company's strategic focus on navigating complex regulatory and commercialization challenges in the rare disease space.

ABVC BioPharma Reports 230% Licensing Revenue Growth and Major Asset Expansion in Q3 2025
ABVC BioPharma achieved significant financial growth with 230% year-over-year licensing revenue increase and 181% asset expansion, driven by strategic investments in Taiwan manufacturing facilities that strengthen the company's global dual-core operational structure.

Cancer Treatment Support Needs Drive Biotech Innovation
The emotional and physical challenges faced by cancer patients during treatment are motivating biotech companies like Soligenix to develop new therapeutic approaches while highlighting the importance of patient-focused support strategies.

Alpha Cognition Secures $35 Million in Oversubscribed Public Offering to Accelerate Neurodegenerative Disease Treatment Commercialization
Alpha Cognition's successful $35 million public offering will accelerate commercialization of its Alzheimer's treatment ZUNVEYL and advance development of other neurodegenerative disease therapies, addressing critical unmet medical needs.

Oncotelic Therapeutics CEO Outlines Key Clinical Milestones in BioMedWire Podcast Interview
Oncotelic Therapeutics CEO Dr. Vuong Trieu discussed the company's upcoming clinical and regulatory milestones in oncology and rare disease treatments during a recent podcast appearance, highlighting potential catalysts for investor attention.

Oncotelic Therapeutics Advances Late-Stage Oncology Pipeline Under Proven Leadership
Oncotelic Therapeutics is progressing multiple late-stage clinical programs in high-unmet-need oncology indications under CEO Dr. Vuong Trieu, whose track record with blockbuster cancer drugs positions the company for potential therapeutic breakthroughs.

Oncotelic Therapeutics CEO's Patent Portfolio and Track Record Position Company for Oncology Market Growth
Oncotelic Therapeutics' CEO Dr. Vuong Trieu, developer of Abraxane, brings extensive patent expertise and successful drug development experience that strengthens the company's position in advancing innovative cancer therapies with significant market potential.

Quantum BioPharma Receives Health Canada Product License for Qlarity Natural Health Product
Quantum BioPharma Ltd. has secured Health Canada approval for its Qlarity natural health product, expanding its market reach and revenue potential through regulatory clearance for cognitive and energy support supplements.

Nutriband Secures $5.3 Million Through Warrant Exercises, Bolstering AVERSA™ Fentanyl Development
Nutriband Inc. raised $5.3 million through voluntary warrant exercises at $6.43 per share, demonstrating strong investor confidence in its abuse-deterrent transdermal patch technology AVERSA™ Fentanyl ahead of its FDA submission.

Nutriband Reports 51% Revenue Growth, Advances Abuse-Deterrent Fentanyl Patch Toward FDA Approval
Nutriband Inc. achieved significant revenue growth and strengthened its financial position to advance AVERSA Fentanyl, potentially becoming the first global abuse-deterrent transdermal fentanyl product with estimated peak annual sales of $80-200 million.

Quantum BioPharma Receives BUY Rating with $45 Price Target from Kingswood Capital
Quantum BioPharma Ltd. received an independent BUY rating and $45 price target from Kingswood Capital Partners, signaling strong analyst confidence in the company's neurodegenerative disease treatment pipeline and future commercial potential.

Oncotelic Therapeutics Highlights CEO's Track Record and Clinical Pipeline Advancements
Oncotelic Therapeutics showcases CEO Dr. Vuong Trieu's substantial patent portfolio and successful drug development history while updating progress on its oncology and immunotherapy pipeline.

ABVC BioPharma Subsidiary Receives $230,000 Milestone Payment in Botanical Drug Partnership
BioLite, an ABVC BioPharma subsidiary, received a $230,000 milestone payment from AiBtl BioPharma, advancing their collaboration on botanical-derived treatments for depression and ADHD that could generate up to $667 million in total value.

Lantern Pharma Advances Oncology Pipeline with AI-Driven Clinical Progress and Extended Financial Runway
Lantern Pharma reports significant clinical trial advancements and AI platform enhancements while maintaining strong financial discipline, positioning the company for continued oncology drug development through mid-2026.

ABVC BioPharma Receives $100,000 Milestone Payment in Vitargus Licensing Deal
ABVC BioPharma's latest milestone payment from ForSeeCon Eye Corporation strengthens a licensing agreement potentially worth $63.5 million for Vitargus®, positioning the company to capitalize on the multi-billion dollar global vitreous substitute market.

Calidi Biotherapeutics Prices $6 Million Public Offering to Advance Clinical-Stage Cancer Therapies
Calidi Biotherapeutics has priced a $6 million public offering to fund development of its Redtail platform, which uses engineered oncolytic viruses to deliver genetic medicines to metastatic cancer sites, addressing high unmet medical needs in oncology.
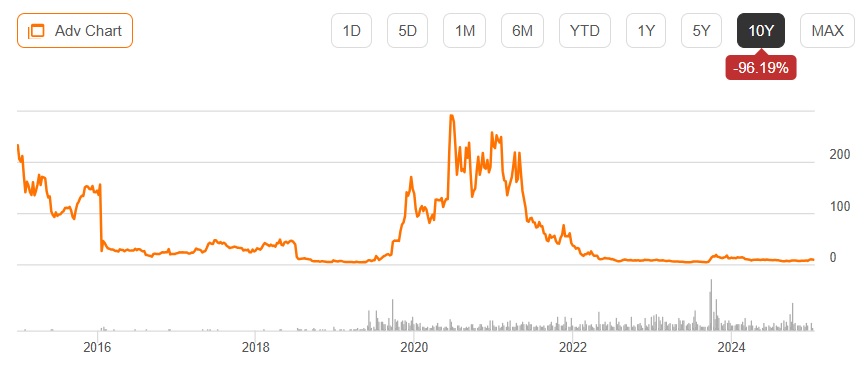
Bank of America Downgrades Novavax to Underperform Citing Long-Term Growth Concerns
Bank of America downgraded Novavax to Underperform due to concerns about the vaccine maker's sustainable growth prospects amid shifting COVID-19 variants and heightened FDA regulatory requirements.

ABVC BioPharma, Inc. Achieves Significant Asset Growth and Secures Licensing Revenue in Q2 2025
ABVC BioPharma, Inc. reports a 103% year-over-year increase in total assets and secures $350K in post-quarter licensing revenue, highlighting strategic growth and operational efficiency.