Pharmaceuticals

Quantum BioPharma Receives BUY Rating with $45 Price Target from Kingswood Capital
Quantum BioPharma Ltd. received an independent BUY rating and $45 price target from Kingswood Capital Partners, signaling strong analyst confidence in the company's neurodegenerative disease treatment pipeline and future commercial potential.

NanoViricides to Present Broad-Spectrum Antiviral Platform at LSX World Congress 2025
NanoViricides will showcase its clinical-stage nanoviricide technology targeting multiple viral diseases at the LSX World Congress, highlighting its potential to address unmet medical needs through a novel antiviral approach.

Nutriband Advances AVERSA™ Fentanyl Patch Toward FDA Submission with Scaled Manufacturing
Nutriband Inc. has scaled manufacturing for its abuse-deterrent fentanyl patch AVERSA™ through collaboration with Kindeva, positioning the company for FDA submission and potential global commercialization with established patent protection across multiple regions.

Soligenix Expands Rare Disease and Public Health Pipeline in Latest Corporate Strategy Update
Soligenix Inc. showcases its dual-focused strategy addressing both rare diseases with unmet medical needs and critical public health threats through its latest corporate presentation, positioning the company as a significant player in specialized therapeutics and biodefense solutions.

OS Therapies Advances Toward 2026 Osteosarcoma Drug Launch with FDA RMAT Designation
OS Therapies Inc. achieves critical regulatory milestones for its OST-HER2 immunotherapy, positioning for potential accelerated approval and U.S. commercial launch in 2026 to address a 40-year treatment gap in osteosarcoma.

Nutriband Showcases AVERSA™ Abuse-Deterrent Technology Addressing Opioid Crisis at Emerging Growth Conference
Nutriband Inc. presented its AVERSA™ abuse-deterrent transdermal technology at the August 2025 Emerging Growth Conference, projecting peak annual sales of $200 million for its fentanyl patch upon FDA approval with potential to exceed $800 million, offering a critical solution to the ongoing opioid epidemic.

New Medication Baxdrostat Shows Promise in Lowering Blood Pressure and Slowing Kidney Disease Progression
The novel medication baxdrostat demonstrated significant reductions in systolic blood pressure and urine albumin levels in patients with chronic kidney disease and uncontrolled hypertension, potentially offering new treatment options for this high-risk population.

Helix BioPharma Abandons GEM Financing Deal After Strategic Review
Helix BioPharma has decided against proceeding with a previously announced equity financing facility with GEM, citing misalignment with its capital strategy and commitment to shareholder value, while continuing to explore alternative funding options for its oncology pipeline.

Soligenix Receives FDA Orphan Drug Designation for Dusquetide in Behçet's Disease Treatment
Soligenix Inc. has secured FDA orphan drug designation for its investigational therapy dusquetide, marking a significant advancement in treating Behçet's disease following promising phase 2a clinical trial results.

Nutriband Secures $5.3 Million Through Warrant Exercises to Advance Abuse-Deterrent Fentanyl Patch Development
Nutriband Inc. has raised $5.3 million from warrant exercises to fund the clinical development and FDA submission of AVERSA Fentanyl, potentially the world's first abuse-deterrent opioid patch with estimated peak annual U.S. sales of $80-200 million.

Clene's CNM-Au8 Shows Promise in Parkinson's Disease Preclinical Study
Clene Inc. presented preclinical data demonstrating its investigational drug CNM-Au8 improves mitochondrial function and reduces inflammation in Parkinson's disease models, potentially offering a new neuroprotective treatment approach.

Cybin Inc. to Present at H.C. Wainwright Global Investment Conference
Cybin Inc. will present at the H.C. Wainwright 27th Annual Global Investment Conference, highlighting its late-stage neuropsychiatry developments including breakthrough therapy-designated treatments for major depressive disorder and generalized anxiety disorder.

Kairos Pharma to Present at H.C. Wainwright Global Investment Conference
Kairos Pharma's participation in the prestigious H.C. Wainwright conference provides crucial visibility for its innovative cancer therapeutic ENV105, which addresses significant unmet medical needs in drug-resistant cancers.

Soligenix CEO to Present at H.C. Wainwright Global Investment Conference
Soligenix Inc.'s CEO presentation at the upcoming investment conference highlights the company's progress in developing treatments for rare diseases and vaccines for public health threats, signaling important advancements for investors and patients.

Scinai Immunotherapeutics Reports Strong First-Half 2025 Results with Revenue Growth and Improved Cash Position
Scinai Immunotherapeutics demonstrated significant financial improvement in the first half of 2025, with revenue growth, reduced losses, and strengthened liquidity, positioning the company for continued advancement of its anti-IL-17 NanoAb program and CDMO business expansion.

Clene Inc. Advances Neurodegenerative Disease Treatment CNM-Au8® Toward Potential FDA Accelerated Approval
Clene Inc. is progressing its oral treatment CNM-Au8® for ALS and multiple sclerosis through late-stage clinical development, with key FDA meetings and regulatory catalysts potentially accelerating commercialization for patients with limited treatment options.

Zevra Therapeutics to Present New MIPLYFFA Data at International Congress, Including Best Poster Award Winner
Zevra Therapeutics will present four posters on MIPLYFFA (arimoclomol) at the International Congress of Inborn Errors of Metabolism, including a Best Poster award-winning presentation detailing its mechanism of action for treating Niemann-Pick disease type C, highlighting significant advancements in rare disease treatment.

ABVC BioPharma Receives $450,000 in Q3 Licensing Payments from OncoX Partnership
ABVC BioPharma's receipt of $450,000 in licensing payments from OncoX BioPharma highlights the financial strength of their oncology partnership and positions the company to capitalize on the growing $469 billion cancer therapeutics market.

CNS Pharmaceuticals CEO Outlines Glioblastoma Treatment Strategy and Market Opportunities in Podcast Interview
CNS Pharmaceuticals CEO John Climaco discussed the company's progress in developing novel brain cancer treatments and expanding into aggressive cancers like triple-negative breast cancer with brain metastases during a recent BioMedWire podcast appearance.

ABVC BioPharma Subsidiary Receives $230,000 Milestone Payment in Botanical Drug Partnership
BioLite, an ABVC BioPharma subsidiary, received a $230,000 milestone payment from AiBtl BioPharma, advancing their collaboration on botanical-derived treatments for depression and ADHD that could generate up to $667 million in total value.

NRx Pharmaceuticals' Depression Treatment Receives FDA Fast Track Designation
NRx Pharmaceuticals' NRX-100 received FDA Fast Track designation for treating suicidal ideation, potentially expanding its market to 13 million U.S. adults and offering a novel approach targeting neuroplasticity in depression treatment.

ABVC BioPharma Receives $100,000 Milestone Payment in Vitargus Licensing Deal
ABVC BioPharma's latest milestone payment from ForSeeCon Eye Corporation strengthens a licensing agreement potentially worth $63.5 million for Vitargus®, positioning the company to capitalize on the multi-billion dollar global vitreous substitute market.

Quantum BioPharma Announces Debt Settlement, Equity Grants, and Advisor Departure
Quantum BioPharma has settled outstanding debt through share issuance, granted restricted share units tied to regulatory milestones for its Lucid MS program, and terminated an advisor agreement, reflecting strategic financial and clinical development moves.

Vaginal Estrogen Tablets Show No Increased Stroke Risk for Postmenopausal Women with Prior Stroke History
A Danish registry study found vaginal estrogen tablets do not increase recurrent stroke risk in postmenopausal women with prior strokes, offering reassurance for safe symptom management in this high-risk population.

ReAlta Life Sciences Gains EMA Orphan Drug Designation for Graft-Versus-Host Disease Treatment
ReAlta Life Sciences received European Medicines Agency Orphan Drug Designation for its drug pegtarazimod, advancing a novel dual-targeting treatment approach for steroid-refractory acute graft-versus-host disease that addresses critical unmet medical needs.

Nutriband Advances Fentanyl Patch Development with FDA Meeting as CEO Pursues Presidential Nomination
Nutriband Inc. progresses its abuse-deterrent fentanyl patch AVERSA™ through FDA regulatory pathways while maintaining leadership continuity during CEO Gareth Sheridan's temporary departure for Irish presidential candidacy.
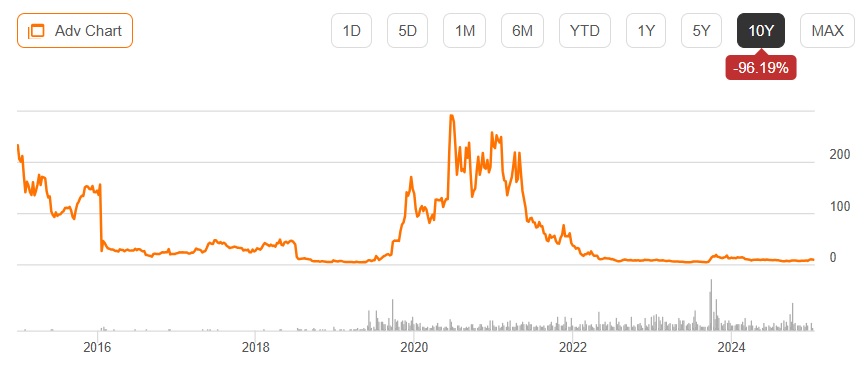
Bank of America Downgrades Novavax to Underperform Citing Long-Term Growth Concerns
Bank of America downgraded Novavax to Underperform due to concerns about the vaccine maker's sustainable growth prospects amid shifting COVID-19 variants and heightened FDA regulatory requirements.

Incannex Healthcare's IHL-42X Shows Promising Phase 2 Results for Sleep Apnea Treatment
Incannex Healthcare Inc. reports significant progress with its IHL-42X treatment for obstructive sleep apnea, achieving up to 83% reduction in apnea events with strong patient-reported outcomes, positioning the company for potential market entry while strengthening its financial position.
BioMed X and Novo Nordisk Collaborate to Revolutionize Oral Peptide Drug Delivery
BioMed X and Novo Nordisk have partnered to tackle the challenge of oral peptide drug delivery, aiming to improve patient compliance and treatment efficacy through innovative formulation technologies.

Nutriband Inc. to Showcase AVERSA(TM) Technology at Emerging Growth Conference
Nutriband Inc. (NASDAQ: NTRB) will present its AVERSA(TM) abuse-deterrent technology at the Emerging Growth Conference, highlighting its potential to revolutionize transdermal pharmaceutical products.